联系人:陈谷一
邮箱:ronniechen@sunano.com.cn
电话:13651969369
地址: 江苏泰州市海陵区江苏省泰州市凤凰西路168号5号楼
Luminosyn? DPP-DTT (also referred to as PDPP2T-TT-OD) is now available featuring:
High molecular weight - higher molecular weight offers higher charge mobility
High purity - DPP-DTT is purified via Soxhlet extraction with methanol, hexane and chlorobenzene under an argon atmosphere
Batch-specific GPC data - so you have confidence in what you are ordering. Also, GPC data is always convenient for your thesis and publications
Large quantity orders - so you can plan your experiments with polymer from the same batch
价格
| Batch
|
Quantity
|
Price
|
|
M315
|
100 mg
|
4800.45
|
|
M315
|
250 mg
|
8011.45
|
|
M315
|
500 mg
|
12635.29
|
|
M315
|
1g
|
20871.50
|
|
M315
|
2g
|
36926.50
|
*for 5 - 10 grams order quantity, the lead time is 4-6 weeks.
Batch Details
| Batch
|
Mw
|
Mn
|
PDI
|
|
M314
|
292,200
|
74,900
|
3.90
|
|
M315
|
278,781
|
76,323
|
3.65
|
General Information
| Synonyms
|
PDBT-co-DTT PTT-DTDPP PDPP-DTT DPPT-TT DPP-TTT PDPP2T-TT PDPP2T-TT-OD DPPDTT Poly[2,5-(2-octyldodecyl)-3,6-diketopyrrolopyrrole-alt-5,5-(2,5-di(thien-2-yl)thieno [3,2-b]thiophene)]
|
|
CAS number
|
1260685-66-2 ()
|
|
Chemical formula
|
(C60H88N2O2S4)n
|
|
HOMO/LUMO
|
HOMO = -5.2 eV, LUMO = -3.5 eV [2]
|
|
Solubility
|
Chloroform, chlorobenzene and dichlorobenzene
|
|
Classification/Family
|
Bithiophene, Thienothiophene, Organic semiconducting materials, Low band-gap polymers, Organic photovoltaics, Polymer solar cells, OFETs
|
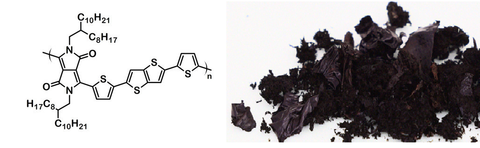
Chemical structure and product image of DPP-DTT, CAS No. 1260685-66-2.
OFET and Sensing Applications
The exceptional high mobility of this polymer of up to 10 cm2/Vs [2] via solution-processed techniques, combined with its intrinsic air stability (even during annealing) has made PDPP2T-TT-OD of significant interest for OFET and sensing purposes.
While the highest mobilities require exceptional molecular weights of around 500 kD (and with commensurate solubility issues), high mobilities in the region of 1-3 cm2/Vs can still be achieved with good solution-processing at around 250 kD. As such, we have made a range of molecular weights available to allow for different processing techniques.
In our own tests, we have found that by using simple spin-coating onto an OTS-treated silicon substrate (using our prefabricated test chips), high mobilities comparable to the literature can be achieved (1-3 cm2/Vs). Further improvements may also be possible with more advanced strain-inducing deposition techniques.
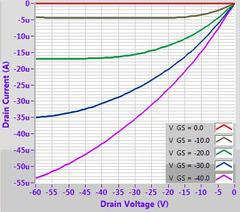
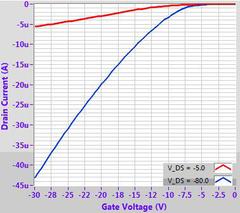
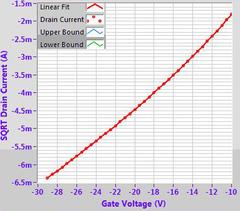
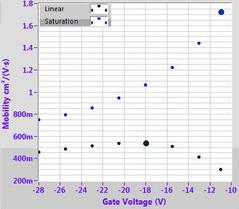
Example OFET characteristics for DPP-DTT (M313) solution processed from chlorobenzene on a 300 nm SiO2 substrate treated with OTS. Output characteristic (top left), transfer curves (top right), mobility fitting (bottom left) and calculated mobility (bottom right).
Photovoltaic Applications
Although shown as a promising hole-mobility polymer for OFETs, when used as the donor material in a bulk heterojunction photovoltaic (with PC70BM as the acceptor), initial efficiencies of 1.6% were achieved for DPP-DTT [3]. The low device metrics were attributed to poor film morphology. However, a higher efficiency of 6.9% was achieved by using thicker film (220 nm) [4].
PDPP2T-TT-OD has also recently been used successfully as an active-layer dopant material in PTB7-based devices [5]. An improvement in device performance was observed, with average efficiencies increasing from 7.6% to 8.3% when the dopant concentration of DPP-DTT was 1 wt%. The use of DPP-DTT as a high-mobility hole-interface layer for perovskite hybrid devices has also been investigated [6].
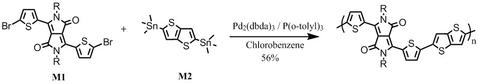
Synthetic route
DPP-DTT synthesis: DPP-DTT was synthesised by following the procedures described in [2] and [3] (please refer to the following references):
With 2-thiophenecarbonitrile and dimethyl succinate as starting materials in t-amyl alcohol, it gave 3,6-Dithiophen-2-yl-2,5-dihydropyrrolo[3,4-c]pyrrole-1,4-dione. Alkylation of 3,6-Dithiophen-2-yl-2,5-dihydropyrrolo[3,4-c]pyrrole-1,4-dione with 2-octyldodecylbromide in dimethylformamide afforded 3,6-bis(thiophen-2-yl)-2,5-bis(2-octyldodecyl)pyrrolo[3,4-c]pyrrole-1,4(2H,5H)-dione. Further bromination gave 3,6-bis(5-bromothiophen-2-yl)-2,5-bis(2-octyldodecyl)pyrrolo[3,4-c]pyrrole-1,4(2H,5H)-dione (M1).
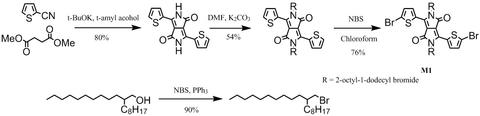
Further reaction of M1 with 2,5-bis(trimethylstannyl)thieno[3,2-b]thiophene (M2) under Stille coupling conditions gave the target polymer DPP-DTT, which was further purified via Soxhlet extraction with methanol, hexane and then chloroform.